Parent Category:
Medical Surveillance of Search Dogs Deployed to the World Trade Center and Pentagon: 2001–2006
ABSTRACT In response to the terrorist attacks of September 11, 2001, at the World Trade Center and Pentagon, almost 50,000 rescue workers and approximately 300 search and rescue dogs participated in rescue and recovery operations. The dogs were exposed to the same hazards as the human workers, but did not have any of the personal protective gear. This prospective double cohort observational study compared annual medical history, blood biochemical and hematologic results, and thoracic radiographic findings in 95 search and rescue dogs that responded to the terrorist attacks at the World Trade Center or the Pentagon on September 11, 2001, to a control group of 55 search and rescue dogs that were not involved in the 9/11 response. Compared to controls, the deployed search dogs demonstrated mild changes in blood work and a higher incidence of radiographic cardiac abnormalities. Species differences may explain the lack of pulmonary findings in the dogs. These dogs may provide early evidence of nonpulmonary complications of the 9/11 response. Continued surveillance of all responders is warranted.
Introduction
The September 11, 2001, terrorist attacks on the World Trade Center (WTC) resulted in over one million tons of debris covering 16 acres and massive clouds of particulates and toxins. Numerous environmental hazards including particulate matter, asbestos, polycyclic aromatic hydrocarbons, metal compounds, dioxins, and volatile organic compounds were identified at the WTC site (Banauch, Dhala, & Prezant, 2005). Acute and delayed respiratory symptoms have afflicted the WTC emergency responders (Moline, Herbert, & Nguyen, 2006; Moscato & Yacoub, 2007; Reissman & Howard, 2008).
The Pentagon attack site was smaller and lacked the hazards associated with the massive crushing and combustion of the WTC but still posed a potential risk for responders. A U.S. Environmental Protection Agency (U.S. EPA) air monitoring summary from October 9, 2001, reported only trace levels of asbestos, volatile organic compounds, and other chemicals. U.S. EPA did, however, identify high concentrations of arsenic and antimony in the soot and ash (Lyman, 2003). In addition to the estimated 40,000 emergency response personnel at the WTC site and over 8,000 responders at the Pentagon, an estimated 250–300 canines responded, including more than 55 dogs at the Pentagon (Otto, Downend, Serpell, Ziemer, & Saunders, 2004; Slensky, Drobatz, Downend, & Otto, 2004). These dogs served three main purposes: detection (e.g., live victims by search and rescue [S&R] dogs, human remains by cadaver dogs, and explosive devices by bomb dogs), patrol (e.g., site security by police dogs), and mental health support (e.g., therapy dogs). At the Fresh Kills Landfill on Staten Island, dogs were used to assist in locating human remains during the sifting and sorting of WTC debris.
Search dog teams started arriving at the WTC on September 11. None of the dogs from outside of the New York City Police Department or the New York-New Jersey Port Authority were present during the tower collapse. The plume of dust, smoke, and toxic components generated by the collapse of the WTC was a major risk factor for human pulmonary complications. The second-highest risk period for both acute and chronic respiratory symptoms in humans occurred during the first two days following the collapse (Herbert et al., 2006; Prezant et al., 2002). Approximately half of the dogs responding to the WTC arrived within the first two days (Otto et al., 2004). The rain on September 14, 2001, likely helped to at least temporarily reduce the overall airborne level of pollutants in lower Manhattan; however, the rain also may have altered the composition of the dusts to which the responders were exposed. In the immediate work zone (Ground Zero), the unrelenting digging and moving of rubble and the uncontained fires resulted in persistent exposure to airborne toxins and particulates.
Although working dogs arrived at the Fresh Kills Landfill on and after September
| Characteristic | Deployed (n = 95) | Control (n = 55) | p-Value |
|---|---|---|---|
| Age (yrs.) | 5.0 (3.0–7.0) | 4.0 (2.0–6.0) | p = .019 |
| Sex | 54 males 41 females |
31 males 24 females |
p = .909 |
| Breeds represented | 11 pure and 8 mixed | 14 pure and 1 mixed | N/A |
| Weight (kg) | 31.2 ± 7.2 | 32.6 ± 7.7 | p = .294 |
| Geographic distribution | 22 states | 16 states and Canada | N/A |
| Breed | Deployed (n = 95) | Control (n = 55) |
|---|---|---|
| Airedale Terrier | 2 | |
| Australian Cattle Dog | 2 | |
| Australian Shepherd | 4 | |
| Beauceron | 1 | |
| Belgian Malinois | 2 | |
| Belgian Tervuren | 1 | 1 |
| Bloodhound | 7 | 1 |
| Border Collie | 1 | |
| Doberman Pinscher | 1 | |
| English Springer Spaniel | 1 | |
| German Shepherd | 30 | 25 |
| German Short-haired Pointer | 1 | |
| Giant Schnauzer | 1 | |
| Golden Retriever | 12 | 2 |
| Hovawart | 2 | |
| Keeshond | ||
| Labrador | 28 | 12 |
| Louisiana Catahoula Leopard Hound | 1 | |
| Mixed breed | 8 | 1 |
| Newfoundland | 1 | |
| Rottweiler | 1 | 1 |
17, the constant sifting and sorting of debris from both the WTC and pre-existing waste continuously aerosolized particulate matter and toxins. At the Pentagon, where about half of the dogs arrived on September 11, personal protective gear and respiratory protection requirements for the human responders were enforced. The use of respiratory protection at the WTC, particularly in the early days of the response, was variable. Regardless of the site, the S&R dogs were not equipped with respiratory protection and foot protection was only used for a limited number of dogs working the site perimeter.
The manifestation of pulmonary disease in workers responding to the WTC disaster has prompted great concern and speculation about long-term hazards from environmental exposure. The risks and long-term effects of response to the Pentagon (Lyman, 2003) have not had such an obvious manifestation. The S&R dogs shared the exposure risks with the human workers.
Companion animals, particularly dogs, may serve as sentinels of human disease due to several similarities between humans and dogs (van der Schalie et al., 1999), including genetics (Lindblad-Toh et al., 2005), physiology, shared diseases, and a common environment. As dogs age, they can develop cancer, heart failure, and dementia—diseases that take decades to manifest in humans. Given the condensed lifespan of the dog, shared environmental hazards may result in clinical manifestations in dogs long before they appear in humans.
Evaluation of the health of the dogs that responded to and were exposed to the hazards associated with these disasters provides information that may help minimize morbidity in future disasters. Importantly, morbidity or mortality in these dogs may provide valuable information for human health care and prevention. The results of annual evaluations that were initiated in October 2001 and continued through September 2006 as part of a medical surveillance program to monitor these dogs are reported here.
Methods
Dogs and Handlers
Handlers of S&R dogs that were deployed to the WTC, Fresh Kills Landfill, and Pentagon disaster sites were identified and contacted for enrollment in a health and behavioral study of their dogs as previously described (Otto et al., 2004; Slensky et al., 2004). The deployed cohort consisted of the S&R dogs that worked at one of the three disaster sites. Dogs that did not deploy to the 9/11 sites, but had similar S&R background and training, constituted the control cohort. Characteristics of the dogs are provided in Tables 1a and 1b.
Data Collection
Handlers completed consent forms to participate in the study and the forms were in compliance with the Institutional Animal Care and Use Committee and Institutional Review Board (IRB) committees at the University of Pennsylvania. The recruitment period began in October 2001 and ended in June 2002. After the recruitment and initial data collection period of Year 1 that ended on September 10, 2002, subsequent data (surveys and samples) collection periods ran from September 11, 2002, until September 10, 2003 (Year 2); September 11, 2003, until September 10, 2004 (Year 3); September 11, 2004, until September 10, 2005 (Year 4); and September 11, 2005, until September 10, 2006 (Year 5).
Surveys
Information from handlers was collected via survey instruments as previously described (Otto et al., 2004; Slensky et al., 2004). Subsequent health surveys for Years 2 through 5 requested complete contact information for the handler and the dog’s veterinarian as well as medical history for the previous year (or since the last survey was completed). The health surveys included questions about the dog’s current status in search and rescue (i.e., active vs. retired), as well as training and deployment activity during the previous year or since the last survey was completed.
Blood Samples
Due to the international distribution of dogs participating in the study, deployed and control dogs were evaluated annually by their local veterinarian. Blood and serum samples were obtained from the dogs and shipped overnight to the University of Pennsylvania for analysis. Complete blood counts and serum biochemical analyses were performed by the Clinical Pathology Laboratory at the Matthew J. Ryan Veterinary Hospital at the University of Pennsylvania.
Chest Radiographs
Pulmonary function testing is impractical in conscious dogs and not widely available. Therefore assessment of the respiratory system was limited to owner report and complete thoracic radiographs (right and left lateral and ventrodorsal or dorsoventral). Radiographs were obtained annually by the dog’s local veterinarian and shipped to the University of Pennsylvania for evaluation by veterinary radiologists who were blinded to the study groups. Serial analysis of the first five years of radiographs was completed by a board-certified radiologist who was also blinded to the study groups. Each radiograph was scored for abnormalities in four categories: pulmonary, cardiac, musculoskeletal, and other.
Mortality
The cause of death was recorded for any dog that died or was euthanized during the study period. A full necropsy was requested. The gross examination was performed by the attending veterinarian and designated samples were shipped to Michigan State University for histopathologic analysis. If the handler failed to have a necropsy performed, the cause of death was recorded according to the attending veterinarian’s diagnosis or as reported by the handler. The detailed results of the postmortem analysis in dogs that died or were euthanized during the initial fiveyear period have been reported (Fitzgerald, Rumbeiha, Emmett Braselton, Downend, & Otto, 2008).
Statistical Analyses
Health Survey
Fisher’s exact test was used to test for an association between a medical condition and whether or not a dog was deployed after 9/11. Fisher’s exact test was also used to test for an association between a medical problem or surgical procedure and whether or not the dog was deployed. Medical or surgical conditions were then sorted into 17 categories for each dog during the entire study period, and Fisher’s exact test was again used to test for an association. Logistic regression was used to determine if the effect of deployment was significantly related to having a urology problem after controlling for the canine’s age.
Hematology and Chemistry
Mixed linear models were used to evaluate mean differences for each of the hematologic and serologic outcomes and included a random subject effect and fixed effects of age, survey time period, deployed (yes/no), and the interaction of time period and deployment. Data was transformed to a normal distribution if necessary before being included in the model.
Radiograph Scores
For each of the three systemic categories (pulmonary, cardiac, musculoskeletal), the proportions of dogs exhibiting radiographic abnormalities were compared between the deployed and the control groups using the Chi-square test of independence. Fisher’s exact test was used when any expected frequency was less than two, or if more than 20% of the expected frequencies were less than five.
Mortality
The proportion of dogs in each of the groups that died during the study period was compared using the Chi-square test of independence Kaplan-Meier survival analyses were performed to test for homogeneity of survival curves over time for deployment (yes/no) and for the different deployment locations. The differences in length of survival from the deployment dates were determined by using the log rank tests of significance. Proportional hazards regression was performed to determine if an association existed between deployment, age, and survival. Dogs that did not die during the study period, or that were lost to follow up, were considered censored for the proportional hazards regression analysis.
Results
Search Dog Characteristics
Of the 216 deployed individuals that were identified and contacted, complete data was collected prior to the close of the first year of the study on 95 dogs (partial data from two additional dogs were included in the previous report after one year [Otto et al., 2004]). Fifty-five of the 114 control dog handlers that were contacted completed the required data to be included.
The overall survey completion rate decreased over time for both groups. For the first study period, 81.9% of deployed handlers completed surveys. For periods 2–4, the response rates decreased to 64.8%, 67.1%, and 61%, respectively. For the four study periods, control handler survey completion rates were 76.4%, 75.9%, 64.6%, and 70.5%, respectively. All handlers that enrolled during study period 0 were given the opportunity to participate every subsequent year that their dog was alive. Some handlers did not provide complete data for each year (e.g., completed surveys Years 0, 2, and 4 only) and therefore participation rates fluctuated. The overall attrition rate was not different between groups (p = .492).
Of the dogs for which data were provided, the average annual retirement rate was 14% for deployed dogs and 12% for controls. The highest single retirement time was in the first year and the most common cause for retirement was age of the dog. Overall, the median age of retirement was seven years for control dogs and nine years for deployed dogs; this difference did not reach significance. Dogs retired due to age were 10 years old on average.
| Condition | Deployed Dogs (n = 95) | Control dogs (n = 55) | p-Value* |
|---|---|---|---|
| Dermatologic | 17 (17.9%) | 9 (16.4%) | >.999 |
| Gastrointestinal | 26 (27.4%) | 11 (20.0%) | .334 |
| Infectious (vectorborne) | 16 (16.8%) | 4 (7.3%) | .135 |
| Musculoskeletal | 26 (27.4%) | 17 (30.9%) | .709 |
| Oncologic | 18 (18.9%) | 7 (12.7%) | .371 |
| Respiratory | 6 (6.3%) | 1 (1.8%) | .423 |
| Toxicologic | 5 (5.3%) | 4 (7.3%) | .725 |
| Traumatic | 27 (28.4%) | 22 (40.0%) | .153 |
| Urologic | 18 (18.9%) | 3 (5.5%) | .027 |
| * Fisher’s exact test. | |||
Time of Arrival and Location of Deployed Dogs
Since air transportation was limited on September 11, 2001, and the days immediately after, search dog teams from the west (California, Texas) and far south (Florida), although deployed earlier, did not arrive at the WTC until after September 14. Thus the actual number of dogs that were exposed to the environmental hazards is smaller than previously reported. The highest risk period for pulmonary complications was during the time of the building collapse (Prezant et al., 2002); no dog in this study was present at that time. The second-highest risk period at the WTC was considered to be the subsequent two days, or until it rained on September 14. Of the 60 study dogs that searched at the WTC, 28 arrived prior to September 14. The time of highest risk at the Pentagon is not known. The first rain also occurred in Washington, DC, on September 14. Of the 23 study dogs that searched at the Pentagon, nine arrived before September 14. The Fresh Kills Landfill did not start receiving Ground Zero debris until September 12, and all 12 dogs in our study arrived at Fresh Kills after September 14.
The median length of time spent searching was 10 days (Otto et al., 2004), with a reported median of 24 hours of active searching. No difference occurred in median search time across the sites. Although active search time and therefore the most intensive exposure was limited to 24 hours, most WTC S&R teams were at Ground Zero for approximately 12-hour shifts. It can be estimated that these dogs were in a highly contaminated zone for approximately 120 hours.
Self-Reported Medical or Surgical Conditions
Owners were queried annually regarding the occurrence of several medical conditions. Since the surveys were collected at various times throughout the year from dog to dog, each medical condition was summarized at the end of the entire study period as to whether the dog ever had the medical condition since 9/11. This summary indicator was used to compare the deployed dogs to the control dogs for each medical condition. The summary of the medical conditions is for all 150 dogs whose handlers completed the initial survey. Although dogs withdrew from the study over time, no statistically significant differences existed in attrition rates between deployed and control dogs for any of the time periods. As a result of the combined effects of attrition and death, approximately 50% of the original deployed dogs remained at the end of the five-year study period and 56% of the original control dogs remained. All 150 dogs were included in the analysis.
The occurrence of any medical condition or surgical procedure since 9/11 of the previous year or since the last completed survey was determined. The most common categories were then summarized for each dog as to whether they had any medical or surgical condition that fell into one of these categoriesduring the entire study period (Table 2). No significant differences existed between deployed and control for any of the categories listed with the exception of urological problems. More deployed dogs (18.9% [18/95]) had a medical or surgical urological condition during the study period compared to control dogs (5.5% [3/55]) (p = .027). Prior to the deployment period, deployed dogs were significantly more likely to have had lower urinary tract signs specifically (12/95 vs. 1/55, p = .023) or any urologic problem (16/95 vs. 1/55, p = .005) than were control dogs. Excluding the prior-to-deployment period, the incidence of urologic conditions (i.e., lower urinary tract signs, urinary incontinence, or prostatic problems) was not significantly different between deployed or control dogs during the deployment period or in the four subsequent evaluation periods.
Blood Chemistry and Hematology
Statistically higher concentrations of glucose, alkaline phosphatase, and cholesterol were detected in dogs that had been deployed, whereas control dogs had statistically higher serum potassium.
The glucose was outside the normal range (>112 mg/dL) in nine deployed dogs and four control dogs, but no dog had persistently elevated glucose and no dog had a glucose measurement higher than 129 mg/dL. Eleven deployed dogs had elevations in alkaline phosphatase (>174 U/L). Of the 10 deployed dogs with multiple measurements, seven had progressive increases in alkaline phosphatase over the study period. The highest value reported was 1,486 IU from a deployed dog undergoing glucocorticoid treatment for immune mediated hemolytic anemia. Subsequent values were within normal range for that dog. Only two control dogs had elevated alkaline phosphatase values; one dog had a transient elevation during period 2 and the other dog had elevated alkaline phosphatase in period 4. Twenty-two cholesterol measurements in 10 deployed dogs were elevated (>317 mg/dL). One control dog had a single elevated cholesterol measurement. Potassium was greater than the reference range (>4.9 mEq/dL) in 21 samples from 15 deployed dogs and in 28 samples from 21 control dogs. No dog in either group was hypokalemic at any time. The biochemical changes were independent of age.
No dog developed diabetes, and the mild elevations in blood glucose may have been a result of stress associated with blood collection or lack of fasting. The alkaline phosphatase elevations may have been associated with exogenous or endogenous (hyperadrenocorticism) steroids or concurrent hepatic disease. Primary hypercholesterolemia is uncommon in dogs and may have been associated with endocrine disease, especially hypothyroidism (Panciera, 1994). Recommendations were made to further evaluate these dogs; however, they were not tested for endocrine diseases as part of our study. The higher serum potassium values in the control dogs may have been related to artifact from hemolysis; no dog reported clinical signs or medical conditions associated with hyperkalemia. Hematologic findings were not different between deployed and control dogs; however, based on subgroup analysis, white blood cell and neutrophil counts were significantly higher in dogs deployed to the Fresh Kills Landfill compared to the WTC (p < .001 and p = .010, respectively). In addition, neutrophil counts from dogs at the landfill were higher than control dogs (p = .001). The peripheral eosinophil counts, which can be increased in dogs with experimentally induced asthma (Collie, DeBoer, Muggenburg, & Bice, 1997), were actually significantly lower in deployed dogs (Otto et al., 2004).
Radiographs
No significant difference existed in the relative frequency of pulmonary or musculoskeletal lesions in the radiographs of deployed dogs compared to control dogs. Radiographic evidence of pulmonary abnormalities (predominately identified as diffuse bronchial and diffuse bronchointerstitial patterns) were present in 27.8% of deployed dogs versus 25.0% of control dogs (p = .916). Radiographs are an insensitive tool to detect changes in pulmonary function. Although pulmonary function testing was not available, retirement rate from search work was used as a surrogate for evidence of decreased respiratory function and was not different between groups. Allergic disease in dogs typically manifests as skin or ear conditions rather than as pulmonary conditions; however, the incidence of ear and skin conditions was not different between the control and deployed dogs.
Musculoskeletal abnormalities were diagnosed radiographically in 54.2% of deployed dogs and 46.7% on control dogs (p = .572). The most prevalent musculoskeletal injuries were arthritis in the appendicular skeleton and vertebral spondylosis. Of the 125 sets of radiographs that were evaluated, 28 control dogs and 40 deployed dogs had one or both of these musculoskeletal conditions.
Radiographic cardiac abnormalities were significantly more likely to be diagnosed in deployed vs. control dogs (9.5% vs. 0.0%, respectively; Fischer’s exact p = .032). Of the seven deployed dogs with cardiac lesions, five had mild left-sided cardiomegaly with atrial involvement, one had right-sided cardiomegaly, and one had mild generalized cardiomegaly. None of these dogs had evidence of congestive heart failure at the time of radiographs. Of the two dogs with radiographic abnormalities that subsequently died, one had histopathologic evidence of degenerative cardiomyopathy. An additional three dogs, which had incomplete radiologic evaluation, also had histopathologic evidence of cardiomyopathy. No control dogs had histopathologic evidence of cardiomyopathy. The incidence of cardiomyopathy was not significantly different between groups, but the number of cases evaluated is small.
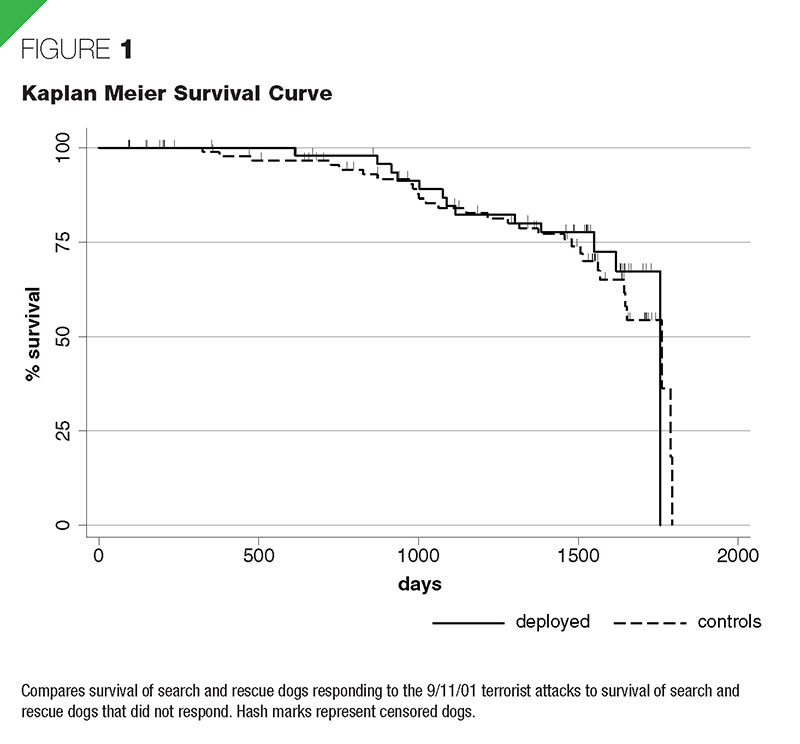
Mortality
The proportion of deployed dogs that died during the follow-up period (31.6% [30/95]) was similar to the proportion of control dogs (23.6% [13/55]) (p = .300). The proportions of deaths at the WTC, Pentagon, and Fresh Kills Landfill were 30.4%, 31.7%, and 33.3%, respectively. The survival curves for deployed and control dogs were not significantly different (p = .639) (Figure 1), nor were they different for deployment locations (p = .919). In multivariate proportional hazards regression, age significantly increased the risk of death (hazard ratio = 1.34; p < .001), but deployment did not (hazard ratio = 0.85; p = .634). The cause of death was assigned to one of five categories; degenerative, neoplasia, infectious/ inflammatory, trauma, and unknown (Figure 2, Table 3). The degenerative group was composed of dogs that the owners elected to euthanize due to progressive deterioration in attitude or perceived quality of life or organ degenerative disease (e.g., cardiomyopathy or degenerative myelopathy). Neoplasia was categorized by the body system affected (Table 3). Both dogs that died of trauma
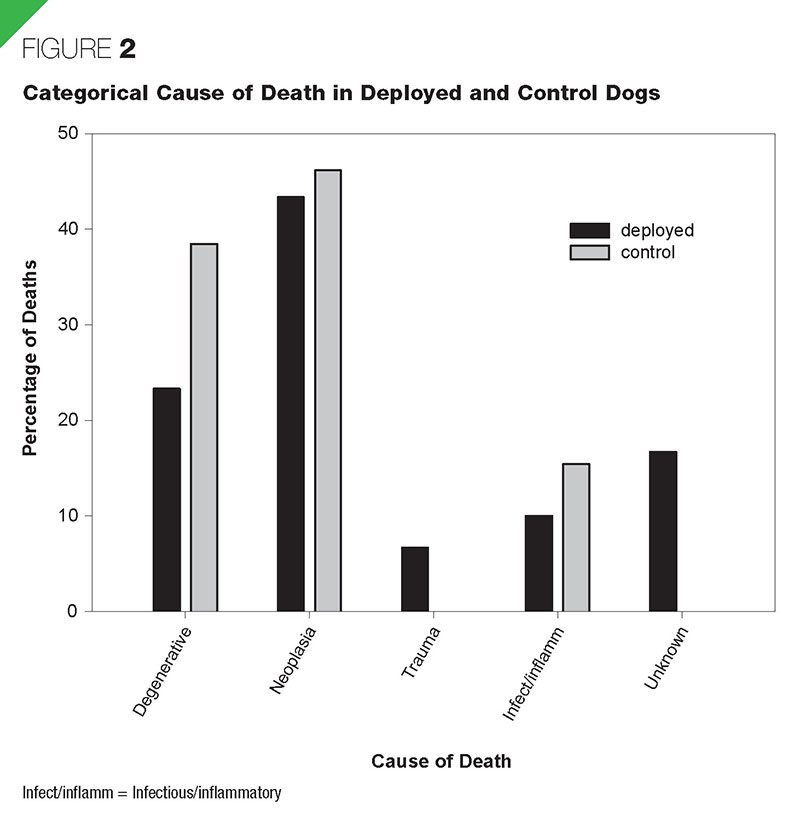
sustained blunt trauma from a motor vehicle. Ischemic disease was categorized in the infectious/ inflammatory category. The unknown category included all cases in which a necropsy was not performed and a premortem diagnosis was unable to categorize the etiology. For example, seizures (n = 1) could have been from degenerative disease, neoplasia, or infectious/inflammatory processes. For all dogs that underwent necropsy, detailed reports of the post mortem analyses has been published elsewhere (Fitzgerald et al., 2008).
Discussion
In these two cohorts of S&R dogs, response to the September 11, 2001, terrorist attacks has not resulted in detectable differences in most parameters studied. No long term follow-up of dogs responding to previous disasters has occurred. After the Oklahoma City bombing, the acute morbidity and short-term effects were reported (Duhaime, Norden, Corso, Mallonee, & Salman, 1998). Four other reports of follow- up in dogs responding to the 9/11 attacks are available. One study reported the acute morbidity associated with the response in 96 S&R dogs from across the country (Slensky et al., 2004). Similar to the acute morbidity reported for human rescue workers, the most common category of complaint was musculoskeletal (Berrios-Torres et al., 2003; Slensky et al., 2004), which included cuts, scrapes, abrasions, sprains, strains, and fractures. Surprisingly, despite the lack of protective gear, only four dogs sustained injuries sufficient to require sutures, and no dog sustained any fractures. In humans, respiratory complaints were the second most common problem, whereas the incidence of respiratory signs in this group of dogs was very low (incidence rate: 2.1 events/1,000 search hours) (Slensky et al., 2004) and was not different across the three sites (WTC, landfill, and Pentagon).
A recent report characterized the acute and long-term morbidity in a group of 27 New York City Police Department (NYPD) dogs that participated in the rescue and recovery response between September 11, 2001, and May 30, 2002 (Fox, Puschner, & Ebel, 2008). This group of dogs did have respiratory and ocular morbidity, which was likely associated with the fact that they arrived at the WTC earlier on September 11 than the group of dogs responding from around the country. Interestingly, the NYPD dogs did not show evidence of respiratory disease over the five years following the response (Fox et al., 2008). In 97 S&R dogs evaluated one year after the response, subtle blood work changes including increased immunoglobulin, bilirubin, and alkaline phosphatase were found in the deployed dogs compared to a cohort of S&R dogs that did not respond to the 9/11 attacks (Otto et al., 2004). Compared to the control dogs, no difference existed in the incidence of medical or surgical problems in the deployed dogs. Neither group had radiographic evidence of pulmonary disease (Otto et al., 2004). In a pathology study of S&R dogs (18 deployed and five control) that died between October 2002 and September 11, 2006, pulmonary anthracosis and particulate matter were commonly found in the lungs of both groups. No primary pulmonary neoplasia was identified in the deployed dogs (Fitzgerald et al., 2008). The most common type of neoplasia in the deployed dogs was hematopoietic (two dogs with lymphosarcoma, one with multiple myeloma, and one with tonsilar squamous cell carcinoma).
The significantly higher incidence of radiographic cardiac abnormalities in deployed dogs was unexpected. Cardiomegaly can result from a variety of causes and can be rightsided, left-sided or generalized. The one deployed dog with generalized cardiomegaly was a Labrador Retriever that eventually died (in 2007) of cardiomyopathy. Dilated cardiomyopathy is a recognized problem in Dobermans, Boxers, and giant breed dogs (Tidholm & Jonsson, 2005). One of the deployed dogs with right-sided cardiomegaly was a Doberman cross that developed cardiomyopathy, which was confirmed on postmortem examination. Three additional deployed dogs without radiographic changes (a Doberman, a German Shepherd, and a German Shepherd cross) were diagnosed on postmortem examination with degenerative cardiomyopathy. No control dogs had either radiographic or postmortem evidence of cardiac abnormalities. Cardiomyopathy in dogs has been attributed to a variety of genetic and environmental factors and could be the manifestation of nutritional deficiencies, immunologic responses, infectious disease, or toxin exposure. Dogs chronically exposed to air pollution had evidence of histopathologic cardiac changes (Calderon-Garciduenas, Gambling et al., 2001) and inhaled particulate matter has been associated with increased cardiovascular morbidity in humans (Brook et al., 2004). Continued surveillance, with attention to cardiac morbidity, is warranted
| Characteristic/Cause | Deployed (n = 30) | Control (n = 13) |
|---|---|---|
| Site | WTC = 19 Landfill = 4 Pentagon = 7 |
N/A |
| Age at death (years) (p = .251) | 9.9 ± 3.0 | 11.1 ± 2.8 |
| n = confirmed by necropsy | n = 15 | n = 5 |
| Degenerative | 8 (n = 6) | 5 (n = 3) |
| Neurologic | 5 | 2 |
| Musculoskeletal | 0 | 3 |
| Cardiac | 3 (one additional dog with a primary diagnosis of neoplasia | 0 |
| Neoplasia | 13 (n = 6) | 6 (n = 2) |
| Hematopoietic | 4 | 0 |
| Vascular | 2 | 1 |
| Urogenital | 2 | 1 |
| Musculoskeletal | 2 | 1 |
| Hepatic/Gastrointestinal | 1 | 1 |
| Skin | 1 (concurrent cardiomyopathy) | 0 |
| Neurologic | 1 | 1 |
| Pulmonary | 0 | 1 |
| Trauma | 2 (n = 1) | 0 |
| Infectious/inflammatory | 2 (n = 2) | 2 (n = 0) |
| Gastrointestinal | 1 | 2 |
| Musculoskeletal | 1 | 0 |
| Unknown | 5 (n = 5) | 0 |
| Neurologic | 3 | |
| Gastrointestinal | 1 | |
| Urogenital | 1 |
The absence of notable long-term morbidity in these dogs, particularly the lack of respiratory signs, is dramatically different than the findings in the human rescue workers. The earliest manifestation was the “WTC cough” (Prezant et al., 2002). Over time, the incidence of reactive airway disease and abnormal pulmonary function has been recognized in up to 70% of workers evaluated (Herbert et al., 2006). The risk associated with pulmonary signs was directly associated with the time of arrival at the WTC (Banauch et al., 2006; Prezant et al., 2002). The early use of respiratory protection was sporadic (Banauch et al., 2006). Regardless of arrival time, the dogs did not have any respiratory protection. During active scenting, dogs breathe through their noses, which may have allowed for better filtering of the large particulates compared to their mouth breathing handlers. Conversely, when dogs pant to maintain their body temperature, dead space (i.e., upper airway) ventilation increases (Robertshaw, 2006), which may have increased exposure of the tracheobronchial region to particulates.
Although not measured, the toxins and particulate matter in the air were thought to be at their highest level during the collapse of the towers. The air quality remained poor with high particulate matter until the rains of September 14 (Lioy & Georgopoulos, 2006). Almost half of the deployed dogs working in New York City arrived at the WTC before September 14, during the period of moderate risk. The Pentagon response has not been associated with physical morbidity.
Several reports cite an increased incidence of cardiovascular morbidity following the 9/11 terrorist attacks (Allegra, Mostashari, Rothman, Milano, & Cochrane, 2005; Feng, Lenihan, Johnson, Karri, & Reddy, 2006; Holman et al., 2008; Ornato, Muller, Froelicher,& Kloner, 2007). These reports attributed the increased incidence of myocardial infarction and other cardiac complaints to increased psychological stress (Allegra et al., 2005; Feng et al., 2006; Holman et al., 2008; Ornato et al., 2007). In dogs, myocardial infarction is rare (Driehuys, Van Winkle, Sammarco, & Drobatz, 1998). Although posttraumatic stress disorder is not recognized in dogs, anxiety can lead to other abnormal canine behaviors (Herron et al., 2008). We have no evidence of an increased incidence of anxiety disorders in the deployed dogs (Otto et al., 2004). The cardiac changes are therefore unlikely related to stress and may portend a previously unrecognized cardiac risk associated with the response to 9/11.
The difference between the respiratory morbidity in the canine and human rescue workers was unexpected, particularly given the high incidence of respiratory signs in the people and the lack of respiratory protection in the dogs. Canine and human pulmonary anatomies are similar, and experimental studies of inhaled particulate matter show similar deposition and clearance patterns in each species (Schlesinger, 1989; Snipes, 1989). In addition, dogs have been used as sentinels of both pulmonary and cardiac effects of chronic exposure to air pollution (Calderon-Garciduenas, Gambling et al., 2001; Calderon-Garciduenas, Mora-Tiscareno et al., 2001). Despite the structural similarities, dogs have relatively larger conduct ing airways and efficient collateral airways that may minimize concentrated exposure to irritants and allergens (Bice, Seagrave, & Green, 2000; Kirschvink & Reinhold, 2008). Dogs are also highly resistant to the development of asthma or reactive airway disease. In experimental settings, dogs have been used as a model of asthma; however, the model requires complex exposure regimens or uniquely bred strains (Hirshman, Malley, & Downes, 1980; Kepron, James, Kirk, Sehon, & Tse, 1977). In these experimental models, airway hyper-reactivity can be documented (Barrett, Rudolph, Bowen, & Bice, 2003), but clinical evidence of pulmonary compromise has not been demonstrated (E.G. Barrett, personal communication, October 31, 2008). In Alaskan sled dogs, cold-induced pulmonary inflammation has been documented in dogs that have completed an 1,100 mile race, suggesting that despite cytologic evidence of inflammation and mucous accumulation, performance was minimally compromised (Davis et al., 2002).
In addition to the resistance to clinical signs of asthma or reactive airway disease, the longer nasal passages and necessity for nasal breathing during scent work may have more effectively filtered the particulate matter and toxins. In the absence of respiratory protection, mouth-breathing human responders were likely to have an increased respiratory accumulation of the larger (>2.5 mm) particles, particularly in the tracheobronchial region (Schlesinger, 1989). The particulate matter at the WTC was predominantly composed of large particles (>10 mm) (Chen & Thurston, 2002). In experimental studies, mouth-breathing humans are more likely than dogs to deposit these large particles in the respiratory tract (particularly in the tracheobronchial tree) (Schlesinger, 1989), providing one potential reason for the difference in pulmonary signs between the species. Dogs also lack many of the complicating behaviors (e.g., smoking). Compared to firefighters, S&R dogs have a much lower occupational hazard, a factor that may exacerbate or contribute to symptoms in people.
Although these S&R dogs do not appear to be sentinels of airway hyper-reactivity, the fact that they shared the same environmental exposure as the human rescue workers suggests that other effects of the response may be evident in the dogs before they are seen in people. The dogs in our study were only exposed for an average of 10 days. While this is a relatively low exposure duration, the majority of dogs were part of urban S&R teams, and the human team members had identical response and exposure times as the dogs. The relatively small number of dogs on this study—95 deployed dogs, with only 73 at the WTC or landfill—would make it impossible to show an association with rare or infrequent conditions that were a result of the exposure.
Although handler reporting is likely to introduce bias, the emphasis on rescue worker pulmonary effects may have made the deployed dog handlers more vigilant in monitoring for respiratory signs. The ideal would have been a more standardized physical examination including pulmonary function testing, but that was not practical.
In conclusion, the dogs that responded to the 9/11 terrorist attacks did not show respiratory symptoms either acutely during deployment or over the subsequent five years. This difference between the human and canine workers is likely a result of difference in both anatomy and physiology. Overall, deployed dogs had minor changes in blood work. Whether these signs were associated with stress, exertion, infection, or toxin clearance cannot be determined. Radiographic evaluation of the heart identified more abnormalities in the deployed dogs versus the controls and four deployed dogs had confirmed cardiomyopathy. The death rate in both groups was similar, as was the diagnosis of cancer. Continued surveillance of all responders is warranted, realizing that the S&R dogs may be sentinels of other or unexpected sequelae to the 9/11 response.
Funding:
This work was supported by grants from the American Kennel Club Canine Health Foundation, Veterinary Pet Insurance, Merial, and FedEx Corporation.
Acknowledgements:
The authors thank Alexis Morris Hubbard for her technical assistance; Drs. Mark Saunders, Lisa Ziemer, and Jennifer Kinns for their radiologic expertise; and Dr. Jennifer A. Taylor for her editorial advice.
Corresponding Author:
Cynthia M. Otto, Associate Professor, Department of Clinical Studies-Philadelphia, University of Pennsylvania, School of Veterinary Medicine, 3900 Delancey St., Philadelphia, PA 19104. E-mail: cmotto@vet.upenn.edu.
References
Allegra, J.R., Mostashari, F., Rothman, J., Milano, P., & Cochrane, D.G. (2005). Cardiac events in New Jersey after the September 11, 2001, terrorist attack. Journal of Urban Health, 82(3), 358–363.
Banauch, G.I., Dhala, A., & Prezant, D.J. (2005). Pulmonary disease in rescue workers at the World Trade Center site. Current Opinion in Pulmonary Medicine, 11(2), 160–168.
Banauch, G.I., Hall, C., Weiden, M., Cohen, H.W., Aldrich, T.K., Christodoulou, V., Arcentales, N., Kelly, K.J., & Prezant, D.J. (2006). Pulmonary function after exposure to the World Trade Center collapse in the New York City Fire Department. American Journal of Respiratory & Critical Care Medicine, 174(3), 312–319.
Barrett, E.G., Rudolph, K., Bowen, L.E., & Bice, D.E. (2003). Parental allergic status influences the risk of developing allergic sensitization and an asthmatic-like phenotype in canine offspring. Immunology, 110(4), 493–500.
Berrios-Torres, S.I., Greenko, J.A., Phillips, M., Miller, J.R., Treadwell, T., & Ikeda, R.M. (2003). World Trade Center rescue worker injury and illness surveillance, New York, 2001. American Journal of Preventive Medicine, 25(2), 79–87.
Bice, D.E., Seagrave, J., & Green, F.H.Y. (2000). Animal models of asthma: Potential usefulness for studying health effects of inhaled particles. Inhalation Toxicology, 12(9), 829–862.
Brook, R.D., Franklin, B., Cascio, W., Hong, Y., Howard, G., Lipsett, M., Luepker, R., Mittleman, M., Samet, J., Smith, S.C., Jr., & Tager, I. (2004). Air pollution and cardiovascular disease: A statement for health care professionals from the expert panel on population and prevention science of the American Heart Association. Circulation, 109(21), 2655–2671.
Calderon-Garciduenas, L., Gambling, T.M., Acuna, H., Garcia, R., Osnaya, N., Monroy, S., Villarreal-Calderon, A., Carson, J., Koren, H.S., & Devlin, R.B. (2001). Canines as sentinel species for assessing chronic exposures to air pollutants: Part 2—Cardiac pathology. Toxicological Sciences, 61(2), 356–367.
Calderon-Garciduenas, L., Mora-Tiscareno, A., Fordham, L.A., Chung, C.J., Garcia, R., Osnaya, N., Hernandez, J., Acuna, H., Gambling, T.M., Villarreal-Calderon, A., Carson, J., Koren, H.S., & Devlin, R.B. (2001). Canines as sentinel species for assessing chronic exposures to air pollutants: Part 1—Respiratory pathology. Toxicological Sciences, 61(2), 342–355.
Chen, L.C., & Thurston, G. (2002). World Trade Center cough. The Lancet, 360(Supplement 1), S37–S38.
Collie, D.D., DeBoer, D.J., Muggenburg, B.A., & Bice, D.E. (1997). Evaluation of association of blood and bronchoalveolar eosinophil numbers and serum total immunoglobulin E concentration with the expression of nonspecific airway reactivity in dogs. American Journal of Veterinary Research, 58(1), 34–39.
Davis, M.S., McKiernan, B., McCullough, S., Nelson, S., Jr., Mandsager, R.E., Willard, M., & Dorsey, K. (2002). Racing Alaskan sled dogs as a model of “ski asthma.” American Journal of Respiratory and Critical Care Medicine, 166(6), 878–882.
Driehuys, S., Van Winkle, T.J., Sammarco, C.D., & Drobatz, K.J. (1998). Myocardial infarction in dogs and cats: 37 cases (1985– 1994). Journal of the American Veterinary Medical Association, 213(10), 1444–1448.
Duhaime, R.A., Norden, D., Corso, B., Mallonee, S., & Salman, M.D. (1998). Injuries and illnesses in working dogs used during the disaster response after the bombing in Oklahoma City. Journal of the American Veterinary Medical Association, 212(8), 1202–1207.
Feng, J., Lenihan, D.J., Johnson, M.M., Karri, V., & Reddy, C.V.R. (2006). Cardiac sequelae in Brooklyn after the September 11 terrorist attacks. Clinical Cardiology, 29(1), 13–17.
Fitzgerald, S.D., Rumbeiha, W.K., Emmett Braselton, W., Downend, A.B., & Otto, C.M. (2008). Pathology and toxicology findings for search-and-rescue dogs deployed to the September 11, 2001, terrorist attack sites: Initial five-year surveillance. Journal of Veterinary Diagnostic Investigation, 20(4), 477–484.
Fox, P.R., Puschner, B., & Ebel, J.G. (2008). Assessment of acute injuries, exposure to environmental toxins, and five-year health surveillance of New York Police Department working dogs following the September 11, 2001, World Trade Center terrorist attack. Journal of the American Veterinary Medical Association, 233(1), 48–59.
Herbert, R., Moline, J., Skloot, G., Metzger, K., Baron, S., Luft, B., Markowitz, S., Udasin, I., Harrison, D., Stein, D., Todd, A., Enright, P., Stellman, J.M., Landrigan, P.J., & Levin, S.M. (2006). The World Trade Center disaster and the health of workers: Five-year assessment of a unique medical screening program. Environmental Health Perspectives, 114(12), 1853–1858.
Herron, M.E., Shofer, F.S., Reisner, I.R., Herron, M.E., Shofer, F.S., & Reisner, I.R. (2008). Retrospective evaluation of the effects of diazepam in dogs with anxiety-related behavior problems. Journal of the American Veterinary Medical Association, 233(9), 1420–1424.
Hirshman, C.A., Malley, A., & Downes, H. (1980). Basenji-Greyhound dog model of asthma: Reactivity to ascaris suum, citric acid, and methacholine. Journal of Applied Physiology, 49(6), 953–957.
Holman, E.A., Silver, R.C., Poulin, M., Andersen, J., Gil-Rivas, V., & McIntosh, D.N. (2008). Terrorism, acute stress, and cardiovascular health: A 3-year national study following the September 11th attacks. Archives of General Psychiatry, 65(1), 73–80.
Kepron, W., James, J.M., Kirk, B., Sehon, A.H., & Tse, K.S. (1977). Canine model for reaginic hypersensitivity and allergic bronchoconstriction. Journal of Allergy and Clinical Immunology, 59(1), 64–69.
Kirschvink, N., & Reinhold, P. (2008). Use of alternative animals as asthma models. Current Drug Targets, 9(6), 470–484.
Lindblad-Toh, K., Wade, C.M., Mikkelsen, T.S., Karlsson, E.K., Jaffe, D.B., Kamal, M., & et al. (2005). Genome sequence, comparative analysis and haplotype structure of the domestic dog. Nature, 438(7069), 803–819.
Lioy, P.J., & Georgopoulos, P. (2006). The anatomy of the exposures that occurred around the World Trade Center site: 9/11 and beyond. Annals of the New York Academy of Sciences, 1076, 54–79.
Lyman, F. (2003). Messages in the dust. What are the lessons of the environmental health response to the terrorist attacks of September 11? Retrieved June 15, 2010, from http://www.neha.org/pdf/messages_ in_the_dust.pdf
Moline, J., Herbert, R., & Nguyen, N. (2006). Health consequences of the September 11 World Trade Center attacks: A review. Cancer Investigation, 24(3), 294–301.
Moscato, G., & Yacoub, M.R. (2007). World Trade Center disaster: Short- and medium-term health outcome. Monaldi Archives for Chest Disease, 67(3), 154–158.
Ornato, J.P., Muller, J.E., Froelicher, E.S., & Kloner, R.A. (2007). Task force II: Indirect and secondary cardiovascular effects of biological terrorism agents and diseases. Circulation, 115(12), 1672–1680.
Otto, C.M., Downend, A.B., Serpell, J.A., Ziemer, L.S., & Saunders, H.M. (2004). Medical and behavioral surveillance of dogs deployed to the World Trade Center and the Pentagon from October 2001 to June 2002. Journal of the American Veterinary Medical Association, 225(6), 861–867.
Panciera, D.L. (1994). Hypothyroidism in dogs: 66 cases (1987– 1992). Journal of the American Veterinary Medical Association, 204(5), 761–767.
Prezant, D.J., Weiden, M., Banauch, G.I., McGuinness, G., Rom, W.N., Aldrich, T.K., & Kelly, K.J. (2002). Cough and bronchial responsiveness in firefighters at the World Trade Center site. New England Journal of Medicine, 347(11), 806–815.
Reissman, D.B., & Howard, J. (2008). Responder safety and health: Preparing for future disasters. Mount Sinai Journal of Medicine, 75(2), 135–141.
Robertshaw, D. (2006). Mechanisms for the control of respiratory evaporative heat loss in panting animals. Journal of Applied Physiology, 101(2), 664–668.
Schlesinger, R.B. (1989). Deposition and clearance of inhaled particles. In R.O. McClellan & R.F. Henderson (Eds.), Concepts in inhalation toxicology (pp. 163–192). New York: Hemisphere Publishing Corporation.
Slensky, K., Drobatz, K., Downend, A., & Otto, C. (2004). Deployment morbidity among search and rescue dogs from 9/11. Journal of the American Veterinary Medical Association, 225(6), 868–873.
Snipes, M.B. (1989). Species comparisons for pulmonary retention of inhaled particles. In R.O. McClellan & R.F. Henderson (Eds.), Concepts in inhalation toxicology (pp. 193–227). New York: Hemisphere Publishing Corporation.
Tidholm, A., & Jonsson, L. (2005). Histologic characterization of canine dilated cardiomyopathy. Veterinary Pathology, 42(1), 1–8.
van der Schalie, W.H., Gardner, H.S., Jr., Bantle, J.A., De Rosa, C.T., Finch, R.A., Reif, J.S., Reuter, R.H., Backer, L.C., Burger, J., Folmar, L.C., & Stokes, W.S. (1999). Animals as sentinels of human health hazards of environmental chemicals. Environmental Health Perspectives, 107(4), 309–315.

